 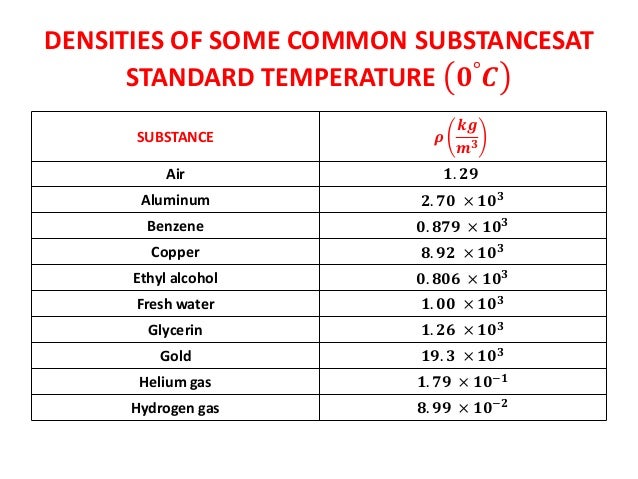
Therefore the overall density of the ship is less than that of the water, causing it to float. A large steel ship also floats on water: Even though steel is denser than water, most of the ship is filled with air, which is less dense than water. For example, oil will float on water because its density is less than that of water. Objects that are less dense float in a liquid that is more dense. The densest naturally occurring chemical element, osmium (atomic number 76), has a density of 22.6 g/cm 3.ĭensity explains why some objects float while others sink. AND AN ESTIMATE OF ITS CRITICAL CONSTANTS1,2. Clearly atoms of gold are heavier than atoms of copper. THE DENSITY OF LIQUID COPPER FROM ITS MELTING POINT (1356K.) TO 2500K. Is the density the same or very close to the density of copper Then the object is made of copper. Weigh the object, then divide by the objects volume to figure out its density. Real copper is fairly dense, measuring at 8.96 g/cm3. For example, the density of gold is 19.3 g/cm 3. Another test that involves math is the density test. The bulk modulus of elasticity of Copper is 140 GPa. 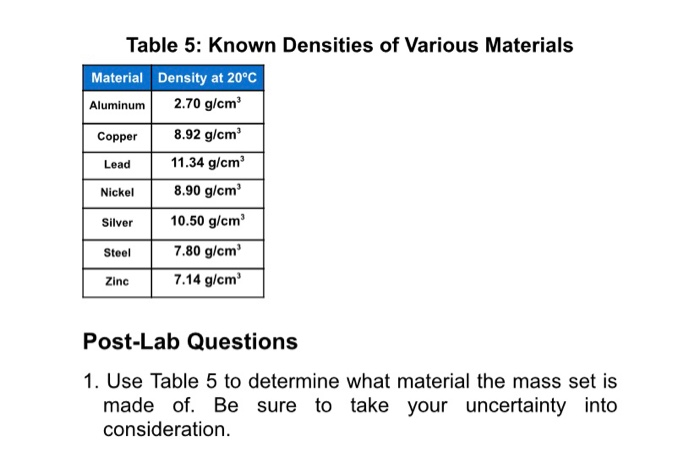
The shear modulus of elasticity of Copper is 48 GPa. Modulus of Elasticity of Copper The Young’s modulus of elasticity of Copper is 33 MPa. Yield Strength of Copper Yield strength of Copper is 33 MPa. Reddish copper alloy (possibly 85 copper and 15 zinc, 8.23 g/cm 3) High purity copper (8. Ultimate tensile strength of Copper is 210 MPa. In solids the particles are closely packed, and so the density is higher. The density of an object or a material is defined as the mass m divided by the volume V in symbols, m/V. The low density of air and other gases can be explained in terms of the large spaces between their particles. For example, the density of air at sea level is 0.0013 g/cm 3, whereas the density of copper metal is 8.96 g/cm 3. The weight of an object, which is usually of more practical interest than its mass, can be determined by multiplying the mass by the acceleration of gravity.ĭensity tells much about how particles are arranged in materials. Density offers a convenient means of obtaining the mass of an object from its volume or vice versa: the mass is equal to the volume multiplied by the density, while the volume is equal to the mass divided by the density. Generally mass is measured in grams (g) and volume in cubic centimeters (cm 3) or milliliters (mL) a cubic centimeter is equal to a milliliter. Virgin Human Hair Hair Color: Natural Black Color Wig Density: 150. The density of any material can be determined by dividing the mass by the volume. Copper Hair Short BobCopper Brown Short Bob Lace Front Wig 10 Human hair blend. To have the same mass of water and rubbing alcohol, a greater volume of alcohol is needed because it is less dense than water. Thus, the density of lead is greater than that of aluminum. A lead block has more mass than the same volume of aluminum. This physical property is defined as the ratio of mass to volume of a substance. The symbol for Dysprosium is Dy and its density g/cm3 is 8.55. Every substance has its own unique value for density. Density Calculations Problems 11-25 Problem 13: Calculate the mass of copper in grams (density 8.96 g/cm3) with the same volume as 100.0 grams of gold (. The symbol for Copper is Cu and its density g/cm3 is 8.96. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
